Is The COVID-19 Vaccine Associated With Increased Joint Inflammation In Patients With Autoimmune Arthritis? A Survey With 1,188 Participants
Clint Paddison (BSc) (info@rheumatoidsolutions.com) , Vidhi Bharti (PhD)
Disclaimer – The content of this analysis is for informational or educational purposes only, and does not provide medical advice.
Introduction
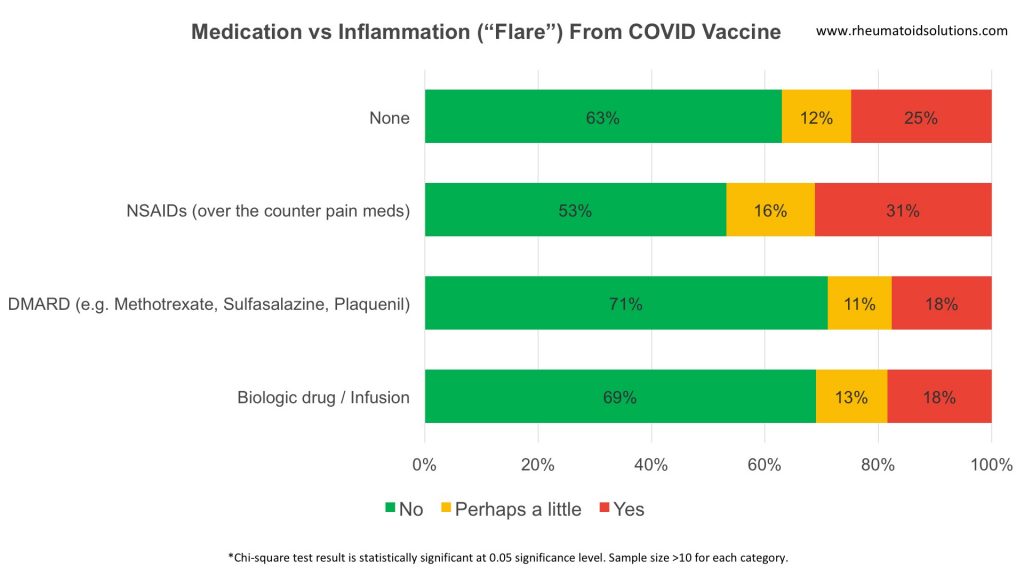
The effectiveness of vaccines against SARS-CoV-2 has been confirmed in real-world studies and available evidence indicates that eligible COVID-19 vaccines have an acceptable short-term safety profile. The most commonly reported systemic side-effects of the vaccine are fatigue and headaches. However, there is limited data to evaluable the possible correlation between these vaccines and joint inflammation in patients with autoimmune arthritis. In this observational study, we present the survey results of 1,188 autoimmune arthritis patients who received one or more COVID vaccine doses and subsequently answered questions relating to their joint inflammation. Of the raw preliminary responses, 20% of patients reported an inflammatory reaction after receiving the vaccine, with an additional 14% reporting a mild inflammatory reaction. Data analysis revealed that NSAID use was associated with a higher incidence of vaccine-induced-inflammatory reaction with statistical significance. Fibromyalgia patients showed a significantly greater incidence of inflammatory reactions, though sample size was small. The was no significant correlation between the type of vaccine, or the duration of illness, with an inflammatory reaction.
Method
A one-page questionnaire was created and distributed via email to 20,338 active subscribers on the Paddison Program mailing list between 17th and 18th August 2021.
The survey questions were:
- What is your diagnosis?
- How long have you had this condition?
- What medications are you taking?
- Which Vaccine did you receive?
- Did you experience inflammatory arthritis symptoms after the vaccine? (e.g. swelling/pain/morning stiffness?)
- If you experienced joint inflammation, how long did these symptoms persist?
- Any other notable side effects or concerns after the vaccine?
Responses from 1,188 patients were received within the allotted 3 day time period.
The results are presented in two parts. The ‘descriptive’ portion which provides simple summaries about the sample and the measures, followed by the data analysis results. In the data analysis, the survey contains the responses as categorical variables. To analyze the relationship between two categorical variables, the chi-square test of association has been used. The test has been performed based on a null hypothesis stating that the two categorical variables are not associated, and an alternative hypothesis stating the two categorical variables are associated. The rejection of the null hypothesis or the failure to reject the null hypothesis has been concluded based on the test statistic, significance level, and p-value.
Results
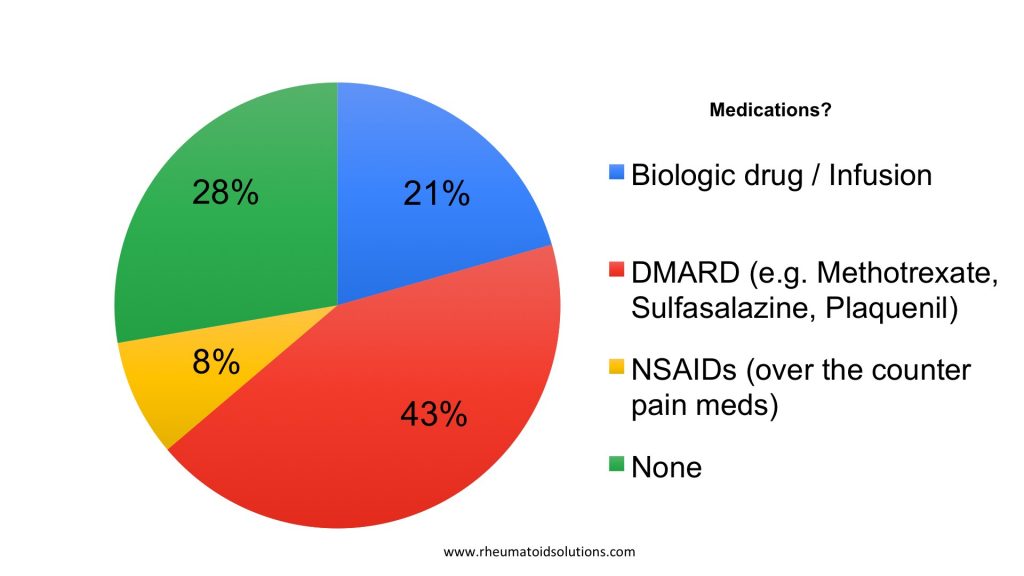
What Medications Are You Taking?
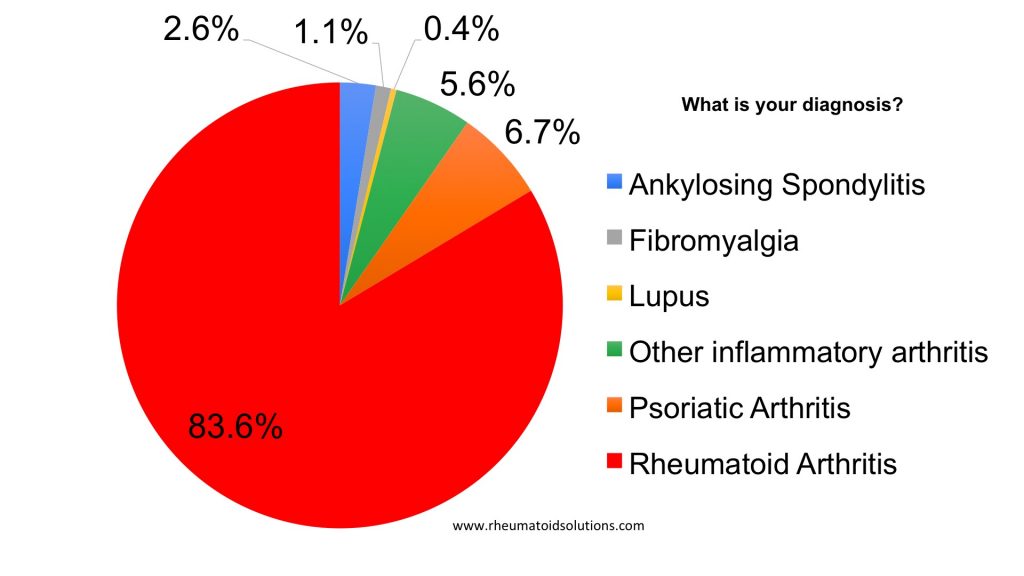
What is your diagnosis?
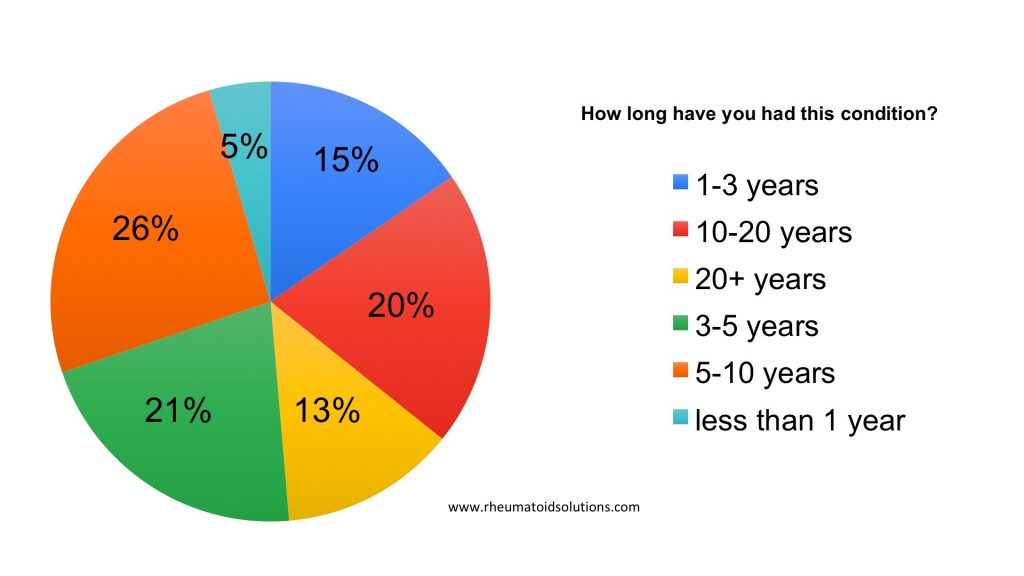
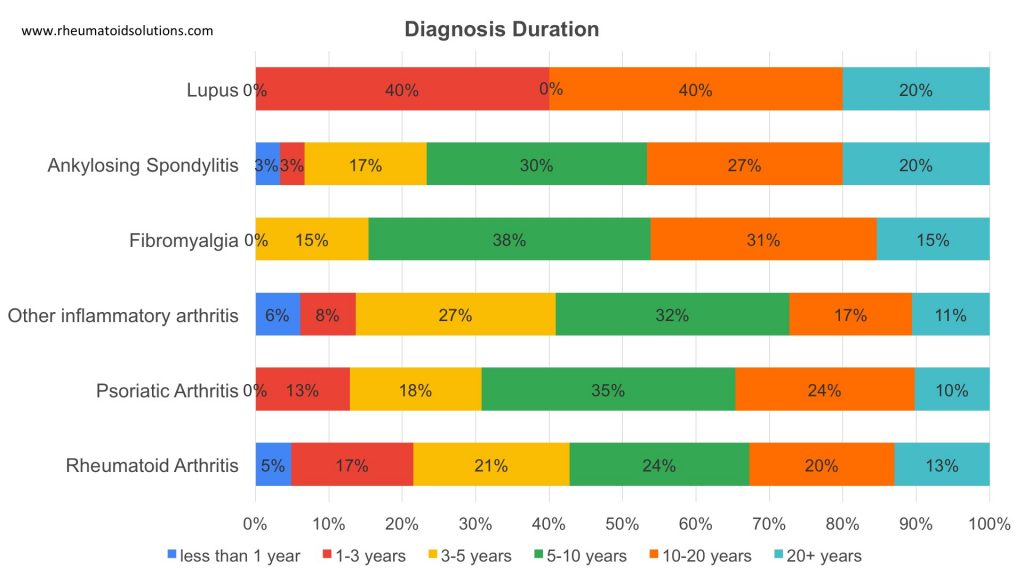
How long have you had this condition?
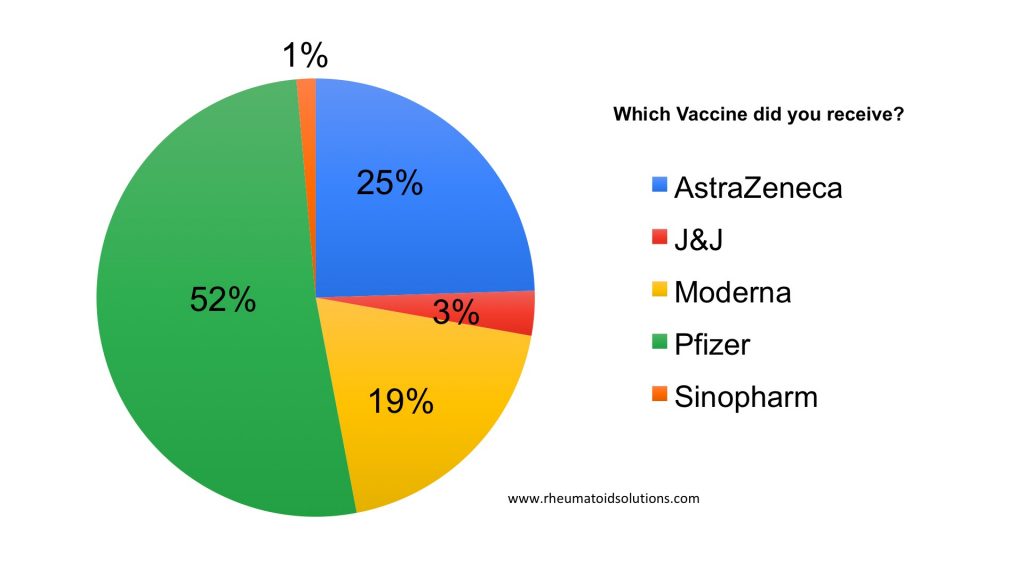
Which Vaccine did you receive?
Did you experience inflammatory arthritis symptoms after the vaccine?
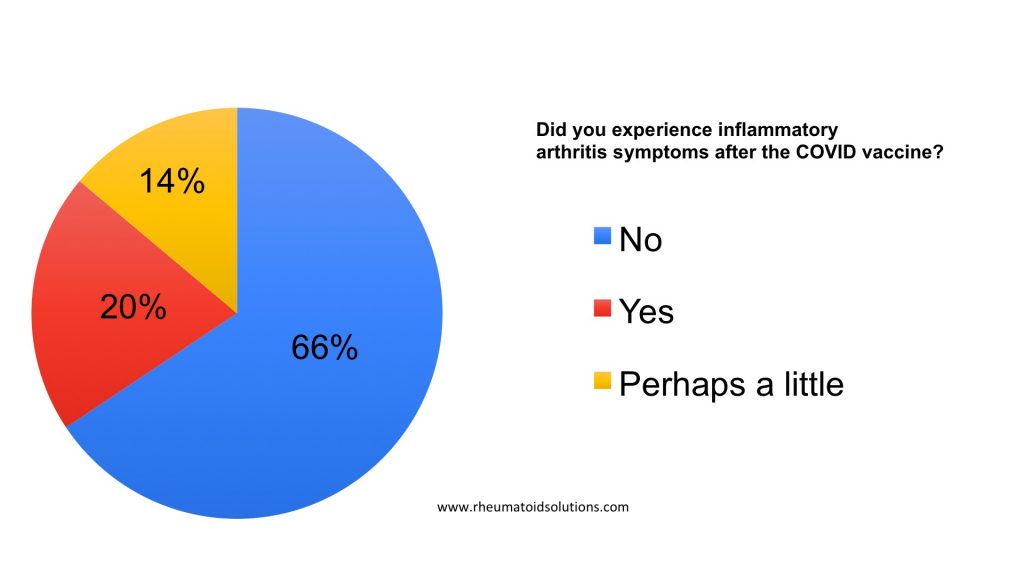
If you experienced joint inflammation, how long did these symptoms persist?
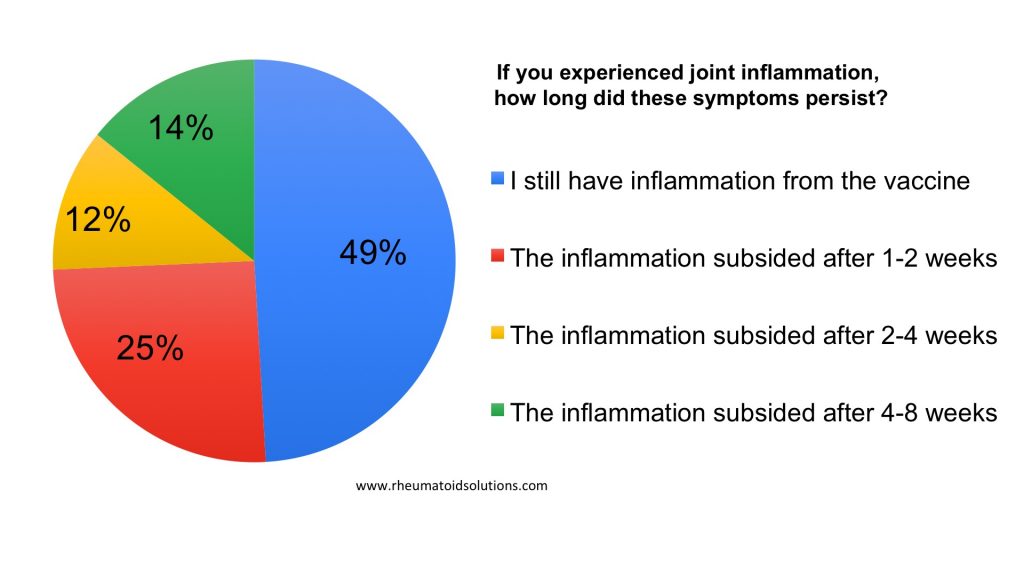
Any other notable side effects or concerns after the vaccine?
Diagnosis duration
Analytical tests were conducted on the data. Results below reveal the vaccine type vs the diagnosis, and the association between the inflammatory response with type of vaccine, diagnosis and medication.
Diagnosis Diagnosis vs Vaccination
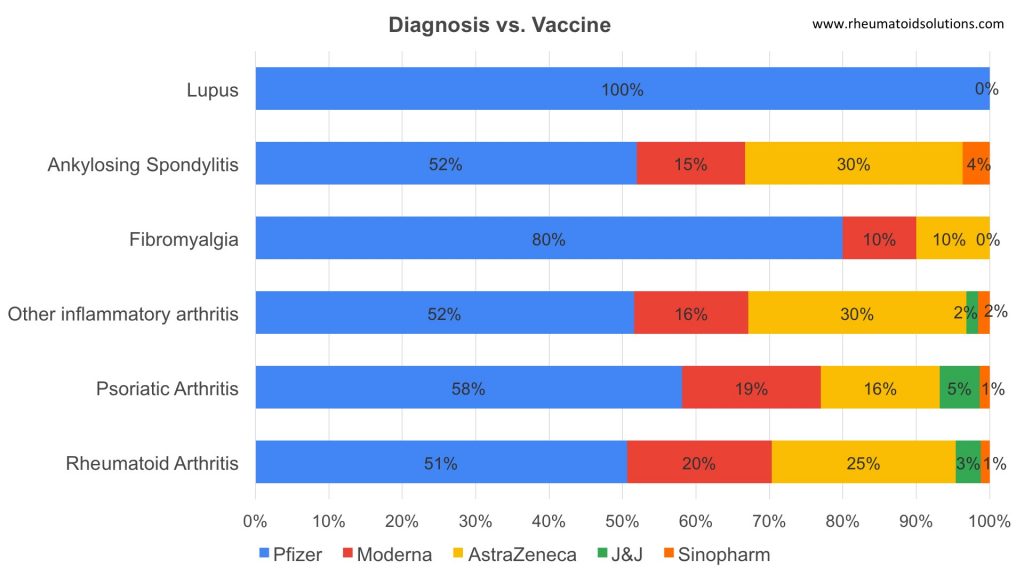
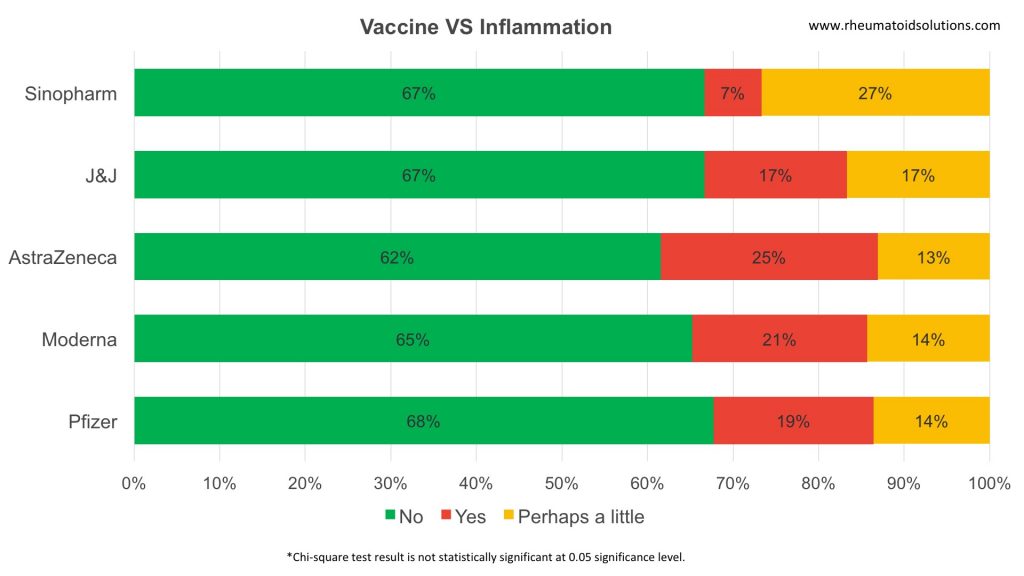
Vaccine vs Inflammation
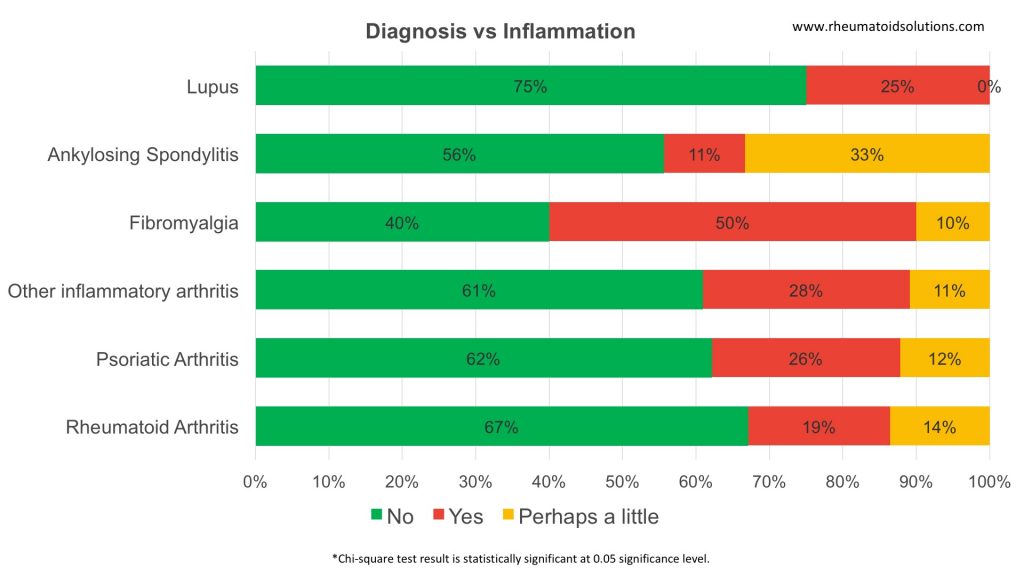
Diagnosis vs Inflammation
Medication Type vs inflammation
Discussion
The number of participants reporting an inflammatory arthritis reaction in this survey was 20%, plus an additional 14% suspecting a possible mild reaction.
By comparison, in the patient-reported GRA Vax survey* 13. 4% of inflammatory arthritis patients reported a flare and 4.6% required new or additional medications.
In the EULAR COVID-19 Vaccination (COVAX) Registry survey of 1,375 entries 5% of patients were documented by practitioners as having a vaccine-induced flare.
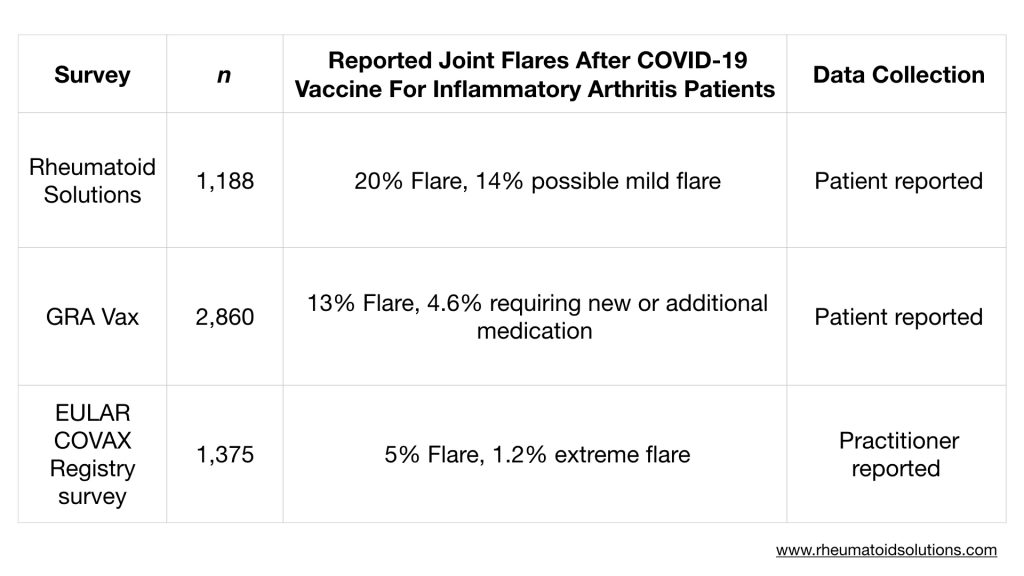
Table 1 reveals that in each survey, a portion of patients with inflammatory arthritis conditions have reported an increase in inflammation. The occurrence of flares reported by patients in the Rheumatoid Solutions and GRA Vax surveys are higher than in the practitioner reported data.
Prednisone should have been included as a drug option since they are a substantial part of the therapeutic arsenal for RA and their accidental omission was an unfortunate oversight. The study would have also benefited from a plant-based diet question, since the Rheumatoid Solutions and Paddison Program audience are often plant-based and patients following a plant-based diet had 73% lower odds of having moderate to severe COVID compared to people who did not follow these diets. It would have been beneficial to see if a plant-based diet reduced the likelihood of an inflammatory response from the COVID vaccine.
There is ambiguity in the answers to the question “If you experienced joint inflammation, how long did these symptoms persist?”. While 51% of survey responders said that their inflammation subsided in 1-8 weeks, the most selected answer of “I still have joint inflammation” does not provide quantitative data as to how long the inflammation has persisted. This question could have been created in such a way to extract more meaningful data.
The answer ‘none’ to the question “Which medications are you taking” is also ambiguous. It may be interpreted by the participant as meaning no medications at all, which was the desired intention, or it may be interpreted as meaning none of the medications listed in the selection.
Conclusion
The statistical analysis suggests a possible association between the medication type and post-vaccine arthritis inflammation. The analysis also suggests a possible relation between the type of diagnosis and post-vaccine arthritis inflammation. This observational study has some limitations. There was not an adequate sample for fibromyalgia and Lupus. No personal attributes such as age, gender or dietary preference were collected. Due to the limited data on this topic, and the widespread interest from autoimmune arthritis patients, a repeat study of this kind would be beneficial, capturing some further data that was no included herein. Of value would be age, gender, diet, how long ago they took vaccine, dose number, and before-vaccine inflammation levels. This would provide a greater understanding of the vaccine-inflammation connection for inflammatory arthritis patients.
* Currently unpublished data, but it is reported at the 29.12 min mark in this presentation organised by the American College of Rheumatology.
Acknowledgments
Thank you to everyone who completed this survey.

Hi Clint. Thank you very much for conducting this survey. This will help a lot when I decide to have myself vaccinated. All your efforts are very much appreciated.
My apologies for not being very participative as I am very busy with work and other concerns. But I try my best to look out for any valuable insights and information from you. God bless you and your family as you are truly a blessing to us who are in this situation.
Warmest regards,
Melissa
Excellent stuff! A limited sample but some interesting indicators in there all the same. Thanks so much for all you do 🙂
Thank you so much to all of you!
Dear Clint,
Thank you very much for this good initiative, nice work.
I have forwarded it immediately to my rheumatologist, I will let you know her possible reaction. You obviously have a precious large address file of motivated people with RA worldwide. Would it not be a good idea to do a follow-up study with more input and data? Interesting for rheumatologists and other scientists. I would be happy to work on that.
Warm regards from Maastricht,
Ted
I
Looking into this now – thank you Ted!
I do have a puzzling question: Why did the nurse tell me not to take advil because it reduces inflammation instead take tylenol. Well if this vaccine needs to show inflammation then how affective could it be for people who are taking medication to reduce inflammation. Also I noticed and reported to you that my CRP had jumped up to 84 6 days after the vaccine. Did anyone report an increase in their CRP.
I thank you for all the hard work that went into the project.
Thank you Clint. Great data. I had mild flu like symptoms and a bit of extra achiness for 36 hours from a 3rd booster shot this week. I'm fine now and good to go! Data is a good thing!
So, interestingly, my remicade is on hold at the moment. It had been 8 weeks since my last infusion when I received my "Moderna" booster. I had no inflammatory response to the prior two vaccines which were timed about 6 weeks after infusion and early on in my infusion treatment. However, with this booster, my arm was warm to the touch, red, swollen and tender. 6 days later, there is still some discoloration and slight tenderness.
Hello Mr.Clint,
Thank you for this survey and explaining the results in easy way.
Now people with any inflammatory diseases can allow themselves to have the Covid-19 vaccine with out fear.
To everyone who tries to get heal, I wish you all the best and happy healing!!
Please do follow the diet from the Paddison program with out fail.
I am doing it and I already noticed so much change in my condition.
The main change was I am able to do yoga asanas with out pain.
Good Luck !!
Thanks for asking these questions. It will be vital to have data about the long term effects of the Covid vaccines. Can you compare the health outcomes of people in the future between the vaccinated and unvaccinated. Thanks again.
Amazing analysis. So clear. Thank you for making it so clear and visually accessible. I had a booster(3rd Moderna)
a couple of weeks ago. Mild side effects including a bot of a flare it seemed.
Clint thank you for putting together this great information. I told my wife every since I got the JJ shot my joints in my knees swell up an still to this day I am dealing with it.Good thing I am still following the paddison program. it will work its way out i am greatfull of that.Thank you for changing my life for the good Clint THANK YOU THANK YOU THANK YOU!!!!!!!!!!!!!!!!!
Thank you too! What lovely energy you have, keep up the great work Chester!
Thanks for sharing the survey! Our child couldn't participate because she isn't vaccinated. Do you also have data about how much of your audience DIDN'T get vaccinated?
What we can share with you is, that our child is in remission for the past year or so and is following the Paddison diet for the past few years together with Methotrexate. She had c-19 half a year ago and thank God had no flare ups. She only had a runny nose.
Wonderful to hear about your daughter’s remission status. I don’t know percentages of vaccinated vs non vaccinated
Excellent survey and very useful results, thanks Clint! Very surprised to see that there was low inflammation from vaccines. I was expecting the total opposite!
Thanks Ric. Data certainly eases the mind!
Thanks! It helpt me alot
This information is absolutely priceless to me. The level of detail surpassed my expectations.Thank you Clint for making it possible.
it is a good effort to do the survey, to help community to aware of the possible side issue, good job, well done
Hi Clint,
Thanks so much, this has been very helpful, I really appreciate learning these results. Knowledge is power.
You have helped me more than you could ever know with my RA journey. Happily I am not on any medication currently and am hopeful for my future.
Sharon from Canada
So awesome Sharon. I’d love to have you share your story on our podcast if you’re interested. Clint
Wow, this is a valuable tool in my decision making process regarding whether to get the vaccine or not. My fear was that I would likely experience a flare up after recieving the vaccine, but as this study shows, the likelihood is somewhat lower than I expected. Thank you so much!
Thank you Clint for sharing a very informative survey. I am hesitant about having the Pfizer vaccine as my rheumatologist has suggested that a safer one for me might be the Novavax vaccine which is currently not available in NZ. I would be interested in your thoughts. Pfizer is the only vaccine available in NZ and it is unclear if any other vaccines will be made available here. I am 66 and have had rheumatoid for 40 years. I currently take sulfasalazine although my arthritis may have burnt out as I had no active joints when I saw my rheumatologist in February. I follow a vegetarian diet and avoid gluten and dairy.
No data on the Novavax yet. The best data I have on the existing vaccines and their association with RA joint ‘flares’ is above, so best to check with Dr. now.
Clint,
Finally some real research has been done to help the RA patient make an educated decision about the COVID vaccine. After getting on my 6th biologic in order to get my 2 year flare under control, I am sure you understand my reluctance to take a vaccine at this crucial time. Thank you for all your time, cost and efforts put into compiling this information. Also, a big thank you to all the participants.
Thank you all!!!
I really appreciate this information as I have been feeling unsure about getting a vaccination but now feel I can go ahead with much more confidence that all will be well. Thank you Clint for giving me peace of mind.
Just wanted to add one more thing. If anyone is concerned about vaccine effectiveness and wether or not they did the right thing by getting the vaccine, I highly recommend watching Dr Been Medical Lectures on you tube. He goes through all the data from around the world and makes it very clear the vaccines are very good. He also talks about other covid treatments, long covid and it's treatments. No agendas or bias. Just no nonsense reporting on what IS going on. Its easy to be confused in this time of rampant misinformation.
Thanks for doing this survey Clint. Just want to report it's been six weeks since my second jab and my mild flare up is starting to settle. I had very strong reaction to the astra jabs and I have been strict whole foods vegan for 4 yeats now, plus active and daily exercise. My crp is always less than 1 and esr is 2, even before methotrexate, in spite of my RA still not being controlled on MTX. I listened to an immunological talk on YouTube and he said not to be concerned if you have a strong reaction because it means your immune system is working well and should mount a good idea defence agains covid. I hope he's right.
Thanks for all your great work
Kylie
Clint, can you erase the bit about strong reaction to vaccine meaning immune system is working well. I just double checked that and the medical consensus is that if you don't have a strong reaction, it doesn't mean you won't develop a good antibody response. The initial reaction is innate immune system working, antibodies are adaptive immune system and how well one system is working doesn't necessarily correlate with the other.
I don’t see that statement
thanks guys very interesting, I had a gout flare up 1st time in 3 months 2 days after AZ
Fantastic report Clint.
Everyone (whether they have an autoimmune disease or not), is concerned about this vaccination.
I am concerned that I am relying on politicians and the media for my information regarding the outcome and effectiveness of this vaccine. (Considering that there was not the extent of research (compared to tetanus, polio, whooping cough etc vaccines), done before we were offered it).
I am definitely more at ease about having 'the jab' after reading your survey.
I wonder though, whether this will have to be an annual vaccine I guess the politicians will give me more feedback once they have reached the important 80% vaccination rate
Tina in Australia
The population in Israel is 80% vaccinated and now there is a huge increase in positive cases and hospitalizations. About 90% of the hospitalized patients and deaths are the 2x vaccinated ones. Now there is a push for the 3rd vaccine and it seems it is fading off quite quickly. And the side effects of the vaccines are increasing as well, including people we know and have spoken to.
The statistics were changed to count those with just 2 jabs as "unvaccinated". Now only the 3x jabbed are considered vaccinated.
I hope this information is helpful, but please do your own research listening to both sides before making a non-reversible decision.
Thank you for sharing your data results with us! I have had the booster shot already and was symptom free after a couple of days. I feel hopeful!
Debra what part of the world are you?
I am in West Australia and we are still
Mostly on our first jab. We don’t have the Covid in our community.
Thanx F
Thank you Clint for caring to put so much work into this survey and for completing it so quickly.
I’ve been waiting for the results with much anticipation…….I need to make a loving decision about Covid vaccination for my brother who is nonverbal and has polymyalgia.
Blessings to you and your family as you encourage and support so many to find health and peace.
Thanks so much
Thank you so much for this study. So much about the RNA/DNA vaccine we just do not know or understand at this point. Those of us who are already struggling with our health have valid concerns about long term effects. The last thing we need is to get worse from a vaccine although none of us want Covid either……
Thanks Clint. Appreciate the effort. I reported possible small RA increase after last jab which was 2 months ago. A nasty flare started 10 days ago ( I’m on Actemra) but is it related to the AZ jab? I’m buggered if I know mate. There are a couple of other factors that may also have kicked it off. Strange disease Clint. I’m 74 had the RA 8 yrs. m also 80 % on your diet.
Thanks to all involved for this very informative survey .Your efforts are so much appreciated .It has given me a little more confidence to get the vacs I’m on MTX and will try to get the Pfizer very soon
Thanks again Clint Paddison, you are such a gem
You’re the best Summar, thank you
Thank you Clint. This was very informative (as are you podcasts). I was diagnosed with RA almost thirty years ago. I have been most fortunate over those years, with minimal meds, several alternatives, good diet, yoga, and a positive attitude, etc. to live a fairly normal life. I had both shots of the moderne vacine, with no adverse reactions. Thanks again for all you do to bring information, tips etc to so many RA folks.
Carol 🙂
Thank you! This is incredibly helpful to see laid out like this.
Wonderful, thanks Sultana, hope you’re doing great
ThankYou Clint for the hard work you’ve put into this survey. Much appreciated ??
Thanks to you, I am drug free & I haven’t had the covid vaccine yet as I’ve had some concerns about how it will affect my inflammation & pain levels. So now I can book it up with some confidence that it will be tolerated within my system.
Warm regards
Marianne ?
Awesome Clint, thank you! My vaccination is due in a couple of weeks, I'm happy to participate in a further study following that. Found this very helpful and would be interested to see the diet detail included. Keep up the great work!
Sensational, and thank you
Very interesting!! Thanks Clint and team
Great work was done Clint. You are so proactive and great boss.
BTW, I hope you are in Florida and not in Australia now.
I did not vaccinate and will not. I don't believe it's vaccine.
Stay safe and best wishes to you and your beautiful family.
Sincerely,
Nailia (Nally) Aleeva
Its important to note that this is not a follow up of how the people will be doing after months pass by when inflammation subsides. This is a survey when vaccines were and are still being rolled out. My daughter is the one with RA. She didn’t feel any flare ups after the first and second dose of the vaccine. But, if temporary flare ups are what people feel after the vaccine, can you imagine what full blown Covid could do?
Thanks Clint for doing this. This took time, effort and money to set this up. It means a lot to everyone.
Thank you so much for conducting this study and sharing the results. I agree that a repeat study with a larger sample size may inform data differences between the available surveys. On a personal note, I followed the guidance provided by the American College of Rheumatology and skipped my dose of methotrexate following each shot. I am curious to know if other survey participants adjusted their autoimmune suppressants as part of their Covid response.
Thank you so much clint for your great efforts. It was very helpful, informative & clearly presented. I was afraid of post vaccine arthritis inflammation. Now i have an idea. I'll get my vaccine Asap.
Very interesting Clint thank you
Jackie
Hi Clint.
Thank you so much.This gives me
Information that I need.Will decide
Soon to get a jab.
Great work, much appreciated.
Mahalo Clint. I am still undecided as I am doing so well and worked so hard to get to where I am today. I don’t want the vaccine or Covid. I am interested in blood type if you would be willing to add that question in.
Interesting. Thanks for this consideration
would love to see a more in depth study.
VERY grateful you've taken this on. I am currently unvaccinated due to an abundance of caution and had a convo with my primary care doc yesterday…she said the jury is out amongst the rheumatologists she has spoken to.
Hi Cling k am taking Rinvoq since January. My first vaccination was scar I gif some stiffness and headaches Siddons flares and shortness of breath but in about 2 weeks I was fine. My second dose was almost no side effects but I was feeling very tired a few days later.
Thanks Clint for doing this survey. Very useful and important information. My dad had an RA flare after the first shot and had to get back on steroid meds. He continued to be on meds for the 2nd shot and didn't see a worsening of RA. I didn't get the vaccine yet for fear it might aggravate my RA as well. I don't take any meds and am mostly WFPB. Thanks for all the work you do.
Very nice information with the graphs and statistics!
Your content is always great!
Thanks for this!
Thanks Clint to you and your team for the valuable work you do.
Thank you!
Thank you for all this work, Clint! As one of the people with PA that experienced a short-term reaction, I was interested to see what others experienced. I personally considered the vaccine worth the reaction and will line up for the booster when it's available. I've seen COVID and I've experienced inflammatory arthritis. You don't want COVID. You really, really, don't want COVID. I'll deal with a flare from the vaccine over that any day.
Clint, this is great information. Many thanks! Proves to me that there is more than coincidence to the timing of my RA flareup and the vaccine.
Thank you Clint for all that you do for others. Having information I can trust means everything:) My RA is not controlled at this time and I take no meds. I have been scared to get the vaccine, and scared not to. Thanks again?
Thank you for the helpful and insightful information and for taking time to produce this survey and the results.
Thank you Clint, and thank you to all those who worked on this study. As an RA patient/warrior, it helps to know that there are professionals working on these kinds of studies. I find the results quite useful personally and I'm sure medical professional will, or at least should, find the results useful to them as well.
Best wishes to all, stay safe!
This information was very helpful in me making a decision to have or not have the vaccine. I have been on a plant based diet for 3.5 years now and don’t take any medication for my Rheumatoid Arthritis for 2 years now. Thank You for the study!
Thx Clint! Having my 2nd shot today after no real reaction to the first Pfizer shot on August 10. I’m off all meds since my last weekly dose on July 28, after a bad liver reaction to MTX. A recent MRI indicated severe liver disease and I’m praying WFPB eating will reverse it. Was told by my RA dr, before the liver problems, to stop MTX a week or 2 before and after the shots. My med change worked that timing out.
I’m having flares since the MTX is now probably out of my system. Rheumy wants to start biologic but my GP is concerned with shutting down the immune system while Covid has just spiked again and been reported to have killed 900 people in Florida in one day.
Prayers welcome for wisdom.
Good job on informing people about vaccine reactions. I thought it was particularly interesting the small difference between those on meds and those on none at all. I read on another site (rawarrior.com) that meds (DMARDs, methotrexate, etc.) only help about 20% of people with RA. Looks like there wasn’t much difference in those groups in your study. Maybe that 20% number is right after all. Thanks for doing the study.
Very genuine study report. ..hats off to Clint paddison for his passionate dedication to help the mankind and serving selflessly in order to help the suffering souls.
Hi Clint,
Great work on this subject.
Personally I decided against getting vaccinated, for a number of reasons.
Just as the vaccines were coming available, our whole family got Covid 19, all at the same time.
We all survived just fine and now have immunity.
Having RA, with being the oldest (almost 70), I was the most vulnerable. My wife, a bit younger totally healthy was less so, and our children even less.
I went back on HydroxyC and zinc and had hardly any of the same symptoms as the rest. They all slept a lot (14 hrs) and were extremely tired. I was not. I only had a mild headache and slept an extra 1/2 hour a day, carrying on my days in quarantine.
I am reading so many articles from around the world about different treatment that works, such as HydroxyC and Ivermectin. Both have been around a long, time, yet so many doctors are unwilling to prescribe it. Take care and keep up the excellent job. Shawn – Happy in Canada
P.S. I am in remission now and off all meds for 9 months….yahoo
I love the last line. Congratulations my friend. Want to do a podcast together? Email me if yes and we’ll set it up
Hi Clint, do you have any experience with children who have JIA that have had the vaccine? Particularly those on Methotrexate and Humira?
I have zero information on this, sorry I can’t help
Hi Anna, my daughter has had JIA and is on MTX, diet and in remission. Not vaccinated. She had covid and it was very mild, just a runny nose. No other symptoms.
I hope this helps.
Very informative study. Thanks for all the work & sharing the results.
Thank you for taking the time to evaluate this! I think it is very helpful. I made the mistake of going off my plant based diet and taking NSAIDs to relieve the pain. Just so happen this was all going on right before I received the vaccination.
Thanks Clint for results from interesting survey and pie graph results.
Im currently participating in a clinical trial being run by my Rheumatologist to see if patients with RA can develop antibodies to either the AstraZeneca or Pfiser Covid vaccinations, by withholding their medications.
Looking forward to hearing of the results!
Hi,
Thanks for doing this study.
Study reported increased % of inflammation after vaccine but so far I know, all the doctors advised to stop arthritis medication for 1/2 weeks before and1/2 weeks after vaccine. And for that reason, arthritis patients who took vaccine in the interval of 21 days, were out of their medication for few weeks and that might be the main reason of the inflammation or pain. I know that this is observational study so you have limitation of the other control factors but you might have added that question in your study.
Great point Farida!
Thank you Clint for the study! It would have also been helpful to include JAK inhibitor drugs such as Xeljanz.
and if medications were help prior to the vaccine. Again, thank you for the study and any further findings you have as we
enter the booster shot phase.
This was encouraging to see this many respond to the questions. It relieved my mind a bit that overall there were no horrible things among large numbers.
Thanks Clint for sharing scientific results after research. I am on your Program but I have to pay after the cheat. I am on a half dose of MTX by my own coz the doctor does not agree with me on the diet concept. Well, I am glad that someone is here who can understand.
Hi Clint
The results are very informative and very useful.
Thank you for taking the time and trouble, it will help people make up their minds one way or the other. Good for people to know that they are not alone too.
Best wishes
Linda
My inflammation started prior to the vaccine and has continued during the past year. I have not contracted Covid and am glad I am vaccinated. If the vaccine impacted my inflammation i would say it was minimal.
Thx for doing this survey, however limited with info like: starting health, diet differences etc.
Sure appreciate your dedication to us all.
I am 61.. have had RA for 9 years.. have flares ( in general) once or twice a year for a month or so.. an on Methatrexate only.. am very active and Eat a Mediterranean diet plus sone homemade low sugar/ avacado oil baked items at times. I had no reaction from P. 2 shots.. just a bit tired for 1/2 day.
Appreciate all you do to inform us?
Laura
Thank you , Clint, for complying this great observational study!
It should read compiling instead of complying . Please edit. Thank you !
Marina
I found this survey interesting ? and very useful. I have fibromyalgia and I did participate in this survey. I took the first of two Pfizer vaccine and because of the joint inflammation I’m seriously considering not taking the second one. I have a doctors appointment next week but my vaccine appointment is this week. The joint inflammation is significant since the vaccine and I couldn’t figure out what was causing it until I participated in this survey.
I will gladly participate in another as suggested above for fibromyalgia and lupus.
Thank you 🙂
Thank you, Clint! Great survey with useful results.
Thank you very much, Clint. I have not received the vaccine yet due to the many unanswered questions about side effects! Your survey has given me the information that I have been searching for!
Great work Clint, thanks very much for your input, I haven’t started your programme just yet but have had a large flare up after having the AZ second jab and had to start a short 3 week course of steroids to help. They have worked so far. Hope they stop it completely. I’ll be starting your programme imminently. I’m trying to get off taking ibuprofen and hydroxychloroquine
Thank you so much! I was waiting for numbers like these for months. They might help me to find an answer to the hard vaccine question as someone with RA without medication. Love from Switzerland, Petra
Thank you Clint, I'm still waiting for my vaccine, but I don't expect to have a problem,.
I've been off Actemra for a year now, that my dietary issues were balanced.
If I'm naughty, I do have a return of inflamation. Mine is GCA and PMR
So far, so good .
I appreciate your wonderful work
Thank you
Judy
I have GCA This is the first time I have read someone else with GCA and say Eating the right food effect how we feel. I have had both injections with no problem but going through a hard time at the moment with terrible head aches and also realise I have been breaking off my stricked eating Habits No Sugar,No Meat,No Dairy, No Gluten . I have read many articles and tried to find doctor or a rheumatologist to believe me that food makes a big difference But all I get told is increase the steroids back up to 50 That is the only way to fix it You have reaffirmed my believe that food makes a big difference Thank You
Thank you so much Clint for doing all this amazing work! Very Insight full!
With all the uncertainty and questions floating around today, it was nice to get some real-time insight from others who are in the same boat as me. Very informative and clearly presented. Thanks Clint!
Comprehensive report, thx Clint.
I meant to write earlier and say that I experienced a flare up a week after my second dose, 16 August.
Your survey came out a day or so after my 2nd dose so I reported no reaction. My flare started on the 23 August & still hasn’t completely settled but is better than it was.
I also wanted to comment on the none reply to the question about medication. For me it meant exactly that- I am in remission & have not had any meds for 17 months.
Kind regards
Annette Le Breton
Very interesting results! I’m going to share this with my rheumatologist! Thanks for putting all of this together and sharing, Clint!
Thank you Jessica
Thank you Clint for you brilliant survey I enjoyed the results
Thank you Marty
Thank you for doing all this work! Great information! I filled out the survey and didn’t have any side effects or inflammation for the vaccine.
Thank you so much for undertaking this study, it's helped solidify my decision not to take part in the ongoing clinical trial for gene therapy.
Well done Clint! So so useful! For people's information I had a terrible and very visible long term response to the Astra Zenaca, (initially saw a lot of swelling in various joints then settled firmly in an already swollen knee after 48 hours and knee became very enlarged for weeks). This finally appears to have settled thanks to a knee joint injection under ultrasound. I'm walking OK at the mo and swelling mostly down.
Thankyou so much for organising this analysis Clint. Very interesting.
I too experienced some extra inflammation after my first shot which has subsequently made me delay having my second (although I will certainly have it)
Thanks again.
Thank you very much for the very informative survey results, Clint. You and your team have put in a lot of hard work on such a fluid topic where the variables keep changing.
I learned a lot and was surprised that quite a few patients had had some degree of pain after the vaccine although not significant in %.
It was also very encouraging to hear that more than 70% of patients on WFPB diet had less severe symptoms of covid. Thank you so much again.
Very useful Clint, easy to read and digest, thank you very much.
Thanks for compiling this data clint & team. Insightful as always. I was in the 20% who had some additional inflammation.
Thank you Richard, sorry to hear about the additional inflammation and I hope it has passed.